Epiontis ID
Robust and consistent clinical immune monitoring
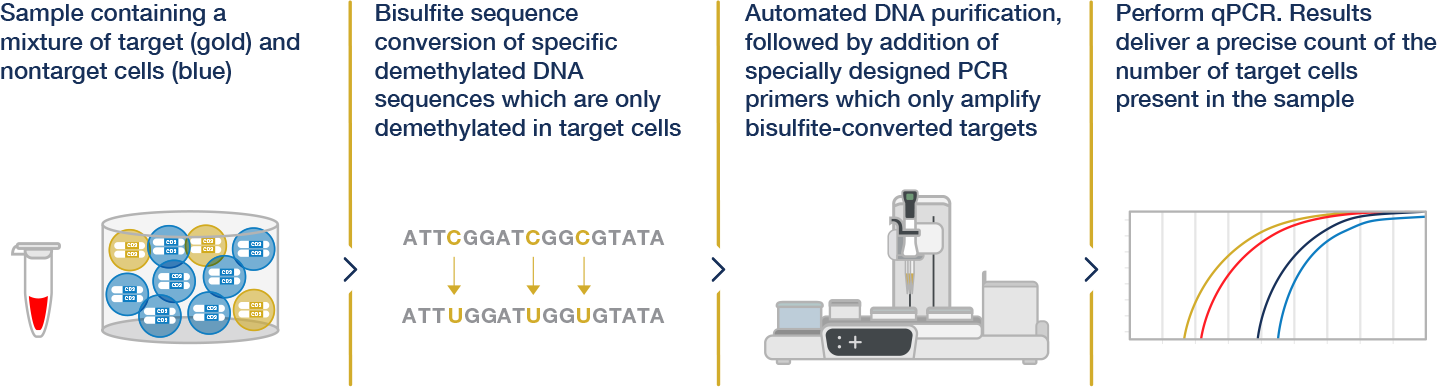
Epiontis ID is an immune monitoring service supporting the development of today’s most innovative therapeutics, allowing researchers to profile and uncover specific changes to the immune system by measuring cell type–specific epigenetic markers in DNA. The stability of DNA enables analysis of varied sample types without complex processing, providing scientists with flexibility in clinical study design, and the automated nature of Epiontis ID produces results that are not subject to operator skill or interpretation.
Scientists can choose any combination of over 30 prevalidated cell types for a customized panel. And because there’s no need for time-intensive validation of each panel, results are delivered rapidly.
Precise and Reproducible Results
Operator-independent technology for consistent results within and across studies
Increased Study Flexibility
Analyze fresh or frozen whole blood, dried blood spots, and even tissue
Simplified Logistics
No need for complex sample preparation or rushed shipments
Rapid Data Delivery
With over 30 prevalidated cell types, data can be delivered within days of project initiation
Proven Clinical Utility
Over 68,000 samples analyzed across more than 100 clinical trials; 17 study sponsor posters and publications
Standardized and Automated Results
Epiontis ID enables accurate counting of cell types by measuring cell type–specific, epigenetic biomarkers using qPCR. The consistency and objective nature of qPCR results lead to data with high reproducibility that can be compared both within and across studies.

About Epiontis ID
Epiontis ID is one of a suite of immune monitoring and biomarker assay solutions offered by Precision for Medicine.
Precision for Medicine is the first global, precision-medicine, clinical research organization. Purpose-built to shift the development curve for life sciences clients, we incorporate laboratory expertise, clinical trial excellence, and advanced data sciences at every stage. Known as Precision Convergence, this integrated approach enables us to deliver critical insights into patient biology from early development through approval. The result: More predictable trial outcomes. Accelerated clinical development. New life-changing treatments for the patients who need them everywhere around the globe.
For Epiontis regulatory information, please visit the Precision for Medicine website regulatory information page.
About Epiontis ID
Epiontis ID is one of a suite of immune monitoring and biomarker assay solutions offered by Precision for Medicine.
Precision for Medicine is the first global, precision-medicine, clinical research organization. Purpose-built to shift the development curve for life sciences clients, we incorporate laboratory expertise, clinical trial excellence, and advanced data sciences at every stage. Known as Precision Convergence, this integrated approach enables us to deliver critical insights into patient biology from early development through approval. The result: More predictable trial outcomes. Accelerated clinical development. New life-changing treatments for the patients who need them everywhere around the globe.
For Epiontis regulatory information, please visit the Precision for Medicine website regulatory information page.
Precision for Medicine
Advancing biomarker-driven trials around the world