Applications of Epiontis ID
With the advent of cell-, gene-, and antibody-based therapies for immune-related diseases and disorders, the role of immune monitoring in both research and clinical care has increased exponentially. Individual parameters, or even comprehensive views, of immune status are central to diagnosing patients, screening eligibility for certain treatments, and predicting treatment response and susceptibility to infection.
Epiontis ID is a particularly ideal tool for supporting the development of autoimmune and immuno-oncology therapeutics, and is ideal for immune profiling needs, including:
Epiontis ID is accredited according to ISO 17025:2018. More information and certifications can be found at the Precision for Medicine web site.

Precise Quantitation of Specific Cell Types
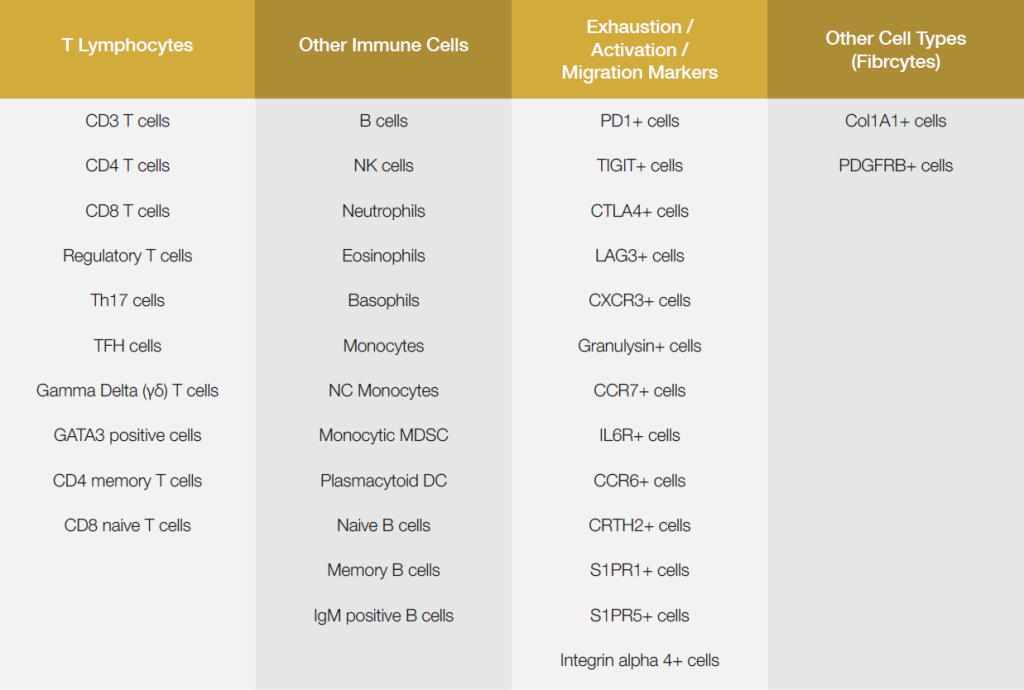
Epiontis ID’s prevalidated assays and ability to deliver rapid results make Epiontis ID an ideal choice for measuring and monitoring specific cell types of interest. Epiontis ID’s prevalidated assays include many cell types crucial for autoimmune indications and for immuno-oncology.
Global, Multicenter Phase 2 and Phase 3 Studies
Epiontis ID is an efficient, economically viable, and convenient global solution for immune monitoring, particularly for large multicenter studies with large numbers of samples involved. Global phase 2 and phase 3 immuno-oncology and autoimmune studies can particularly benefit, as the minimal sample processing and shipping requirements simplify logistics for sites across the globe.


Analysis of Solid Tissues
Immune cell profiling in indications where immune response analysis of solid tissue is needed – for example, autoimmune skin disorders such as psoriasis or salivary gland analysis in Sjogren’s syndrome – are cases where Epiontis ID is ideal. Extraction of viable cells from tissues for flow cytometry can be particularly challenging, but because Epiontis ID can measure nonviable cells, generating reliable results from tissues is much more consistent.
Choose From Any Validated Assay
Currently, there are over 30 validated assays available for Epiontis ID, with more cell types added regularly. Study sponsors can select and combine any of the available cell types into a panel for analysis.
Created in collaboration with Nature, the Epiontis Poster illustrates the numerous cellular pathways involved in autoimmune disorders and their current therapies:

Learn about the benefits and ideal uses of both flow cytometry and Epiontis ID for immune monitoring in clinical studies
